CLINTEL Data Solutions
Transforming Clinical Data
ABL’s Clinical Intelligence (CLINTEL) solution provides a single platform for study analytics. No reconciliation, no more decisions based on inaccurate or incomplete data, no more manipulation of data to make it match reality, no more capturing the same data in multiple systems. Our solution allows your data to work for you instead of you working to access your data. Installation is available in both cloud-based Software-as-a-Service (SaaS), and on-premises configurations. Developed using industry-proven architecture that boasts fast time to implementation, superior visualizations, and technology that is focused on users (e.g. study managers, CRAs, data managers, clinical trial assistants, etc.) as opposed to Information Systems experts.
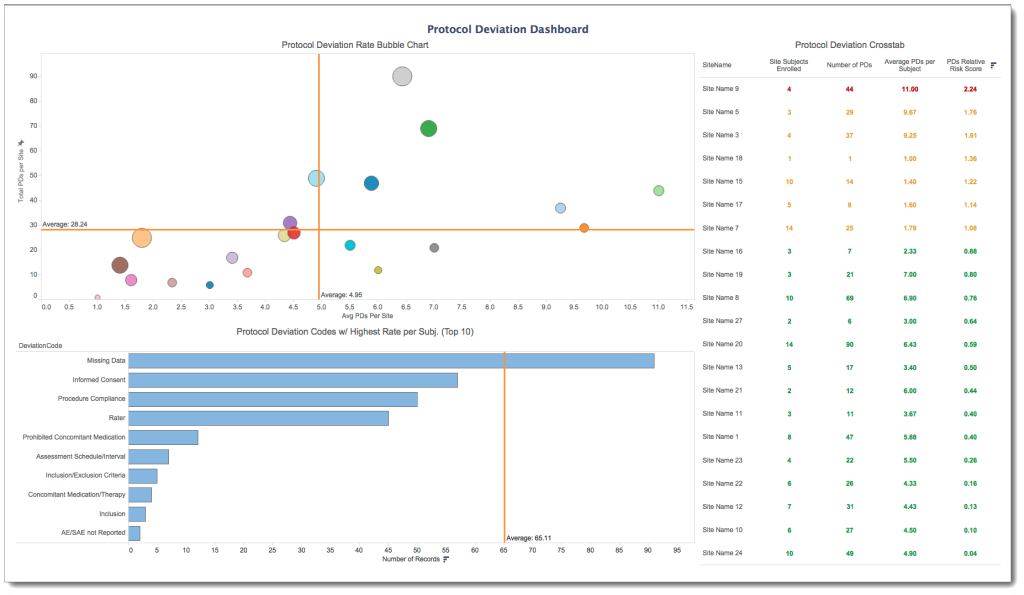
CLINTEL offers a reporting solution that simultaneously accesses data from multiple clinical systems and presents the information in a way that’s geared specifically to development staff working on clinical trials. Our customizable, easy-to-use interface allows non-technical users to view and explore data trends through a series of pre-packaged modules, while also providing users with the ability to create ad-hoc reports and dashboards in minutes. Pre-formatted dashboards are designed to support industry best practices related to risk-based monitoring, data management operational metrics, trial management operational metrics and more. By providing the ability to combine data from multiple data management systems (e.g. Rave®, InForm®, OC RDC®, etc.), clinical trial management systems (e.g. Siebel®, Medidata®, BioClinica®) and safety systems (e.g. Argus®, AERS®, etc.)
Why Choose CLINTEL?
Product Features
We always felt that powerful analytics were only available to companies who had a lot of money to spend on technology. ABL was able to demonstrate that this isn’t the case. They helped us design and implement reports from spreadsheets we received from our CRO’s. Now we have greater insight to our study activities and are able to ensure that our studies are progressing on schedule and on budget.
– Mark R., San Diego, CA